Sbírka 54+ Neutral Atom Of Sodium Zdarma
Sbírka 54+ Neutral Atom Of Sodium Zdarma. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Electron affinity of sodium is 52.8 kj/mol. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Electron affinities reported in unites of kilojoules per mole (kj/mol).
Nejchladnější Study Guide 1 Problems
Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. Therefore, the number of electrons in neutral atom of sodium is 11. Atoms are extremely small, typically around 100 picometers across. 1st ie = 495.8 kj/mol.The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion.
This affinity is known as the. Electron affinities reported in unites of kilojoules per mole (kj/mol). To use electron affinities properly, it is essential to keep track of sign. We will obviously have to find. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. Therefore, the number of electrons in neutral atom of sodium is 11. This affinity is known as the. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom.

Data taken from john emsley, the elements, 3rd edition.oxford: In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: The r max values for neutral gaseous element valence orbitals are abstracted from reference 1.. This affinity is known as the.

They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: 1st ie = 495.8 kj/mol. As the force of attraction is less effective, electrons are removed from the last.. To use electron affinities properly, it is essential to keep track of sign.

Atoms are extremely small, typically around 100 picometers across. Electron affinity of sodium is 52.8 kj/mol.

The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Electron affinity of sodium is 52.8 kj/mol. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. To use electron affinities properly, it is essential to keep track of sign. Atoms are extremely small, typically around 100 picometers across. 1st ie = 495.8 kj/mol. This affinity is known as the. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:.. This affinity is known as the.

To use electron affinities properly, it is essential to keep track of sign. This affinity is known as the. The sodium atom exhibits cation properties.. Atoms are extremely small, typically around 100 picometers across.

As the force of attraction is less effective, electrons are removed from the last. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. As the force of attraction is less effective, electrons are removed from the last. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Electron affinities reported in unites of kilojoules per mole (kj/mol). Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective.

The sodium atom exhibits cation properties. Therefore, the number of electrons in neutral atom of sodium is 11. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. To use electron affinities properly, it is essential to keep track of sign. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. Data taken from john emsley, the elements, 3rd edition.oxford:. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom.

The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:.. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:.

When an electron is added to a neutral atom, energy is released. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. As the force of attraction is less effective, electrons are removed from the last.. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.

In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. The sodium atom exhibits cation properties. When an electron is added to a neutral atom, energy is released. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are extremely small, typically around 100 picometers across. Then play a game to test your ideas! 1st ie = 495.8 kj/mol.. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion.

Therefore, the number of electrons in neutral atom of sodium is 11.. This affinity is known as the. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements.. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.

Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom... Therefore, the number of electrons in neutral atom of sodium is 11. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. As the force of attraction is less effective, electrons are removed from the last. Data taken from john emsley, the elements, 3rd edition.oxford: In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. Then play a game to test your ideas!. The sodium atom exhibits cation properties.

We will obviously have to find.. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. 1st ie = 495.8 kj/mol... When an electron is added to a neutral atom, energy is released.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.. To use electron affinities properly, it is essential to keep track of sign. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. Data taken from john emsley, the elements, 3rd edition.oxford: We will obviously have to find.

Atoms are extremely small, typically around 100 picometers across. Electron affinities reported in unites of kilojoules per mole (kj/mol). In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: To use electron affinities properly, it is essential to keep track of sign. Then play a game to test your ideas! Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. 1st ie = 495.8 kj/mol. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. Therefore, the number of electrons in neutral atom of sodium is 11. As the force of attraction is less effective, electrons are removed from the last. When an electron is added to a neutral atom, energy is released... 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium.

The sodium atom exhibits cation properties. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.

Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective... They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Electron affinity of sodium is 52.8 kj/mol. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1... Electron affinity of sodium is 52.8 kj/mol.

The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion... Data taken from john emsley, the elements, 3rd edition.oxford: 1st ie = 495.8 kj/mol. We will obviously have to find. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. Therefore, the number of electrons in neutral atom of sodium is 11. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Electron affinities reported in unites of kilojoules per mole (kj/mol). They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective.. Data taken from john emsley, the elements, 3rd edition.oxford:

Then play a game to test your ideas! To use electron affinities properly, it is essential to keep track of sign. As the force of attraction is less effective, electrons are removed from the last. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Electron affinities reported in unites of kilojoules per mole (kj/mol). An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.. To use electron affinities properly, it is essential to keep track of sign.

They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. As the force of attraction is less effective, electrons are removed from the last.. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective.

Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements.. 1st ie = 495.8 kj/mol.. Therefore, the number of electrons in neutral atom of sodium is 11.

Then play a game to test your ideas!. Then play a game to test your ideas! The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.

When an electron is added to a neutral atom, energy is released.. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. When an electron is added to a neutral atom, energy is released. Data taken from john emsley, the elements, 3rd edition.oxford: An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. We will obviously have to find. Then play a game to test your ideas! Electron affinity of sodium is 52.8 kj/mol. This affinity is known as the. As the force of attraction is less effective, electrons are removed from the last.

As the force of attraction is less effective, electrons are removed from the last... Atoms are extremely small, typically around 100 picometers across. 1st ie = 495.8 kj/mol. We will obviously have to find. Therefore, the number of electrons in neutral atom of sodium is 11. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Then play a game to test your ideas! As the force of attraction is less effective, electrons are removed from the last. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective.. 1st ie = 495.8 kj/mol.
The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:... The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. When an electron is added to a neutral atom, energy is released. Electron affinities reported in unites of kilojoules per mole (kj/mol). They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The sodium atom exhibits cation properties. Therefore, the number of electrons in neutral atom of sodium is 11. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium.

Therefore, the number of electrons in neutral atom of sodium is 11. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: This affinity is known as the. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Then play a game to test your ideas!

Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective... They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. This affinity is known as the. Atoms are extremely small, typically around 100 picometers across. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. The sodium atom exhibits cation properties.

They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Electron affinities reported in unites of kilojoules per mole (kj/mol). When an electron is added to a neutral atom, energy is released. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium... Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change... Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. This affinity is known as the. Atoms are extremely small, typically around 100 picometers across. Data taken from john emsley, the elements, 3rd edition.oxford: Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1.. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements.

Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: We will obviously have to find. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. As the force of attraction is less effective, electrons are removed from the last. When an electron is added to a neutral atom, energy is released. Atoms are extremely small, typically around 100 picometers across. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. Then play a game to test your ideas!

To use electron affinities properly, it is essential to keep track of sign.. This affinity is known as the. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Atoms are extremely small, typically around 100 picometers across. The sodium atom exhibits cation properties. Data taken from john emsley, the elements, 3rd edition.oxford: 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. Therefore, the number of electrons in neutral atom of sodium is 11. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1.

Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective... We will obviously have to find.. As the force of attraction is less effective, electrons are removed from the last.

1st ie = 495.8 kj/mol. Atoms are extremely small, typically around 100 picometers across. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Electron affinities reported in unites of kilojoules per mole (kj/mol). Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. Therefore, the number of electrons in neutral atom of sodium is 11. To use electron affinities properly, it is essential to keep track of sign.

Therefore, the number of electrons in neutral atom of sodium is 11. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Atoms are extremely small, typically around 100 picometers across. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Data taken from john emsley, the elements, 3rd edition.oxford: When an electron is added to a neutral atom, energy is released. Therefore, the number of electrons in neutral atom of sodium is 11. We will obviously have to find. To use electron affinities properly, it is essential to keep track of sign. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:

When an electron is added to a neutral atom, energy is released. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective.

Atoms are extremely small, typically around 100 picometers across... An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Data taken from john emsley, the elements, 3rd edition.oxford: Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. As the force of attraction is less effective, electrons are removed from the last. To use electron affinities properly, it is essential to keep track of sign. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. When an electron is added to a neutral atom, energy is released. Electron affinities reported in unites of kilojoules per mole (kj/mol).. 1st ie = 495.8 kj/mol.

Then play a game to test your ideas!. This affinity is known as the. Atoms are extremely small, typically around 100 picometers across. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. Therefore, the number of electrons in neutral atom of sodium is 11. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Then play a game to test your ideas! 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium.. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.
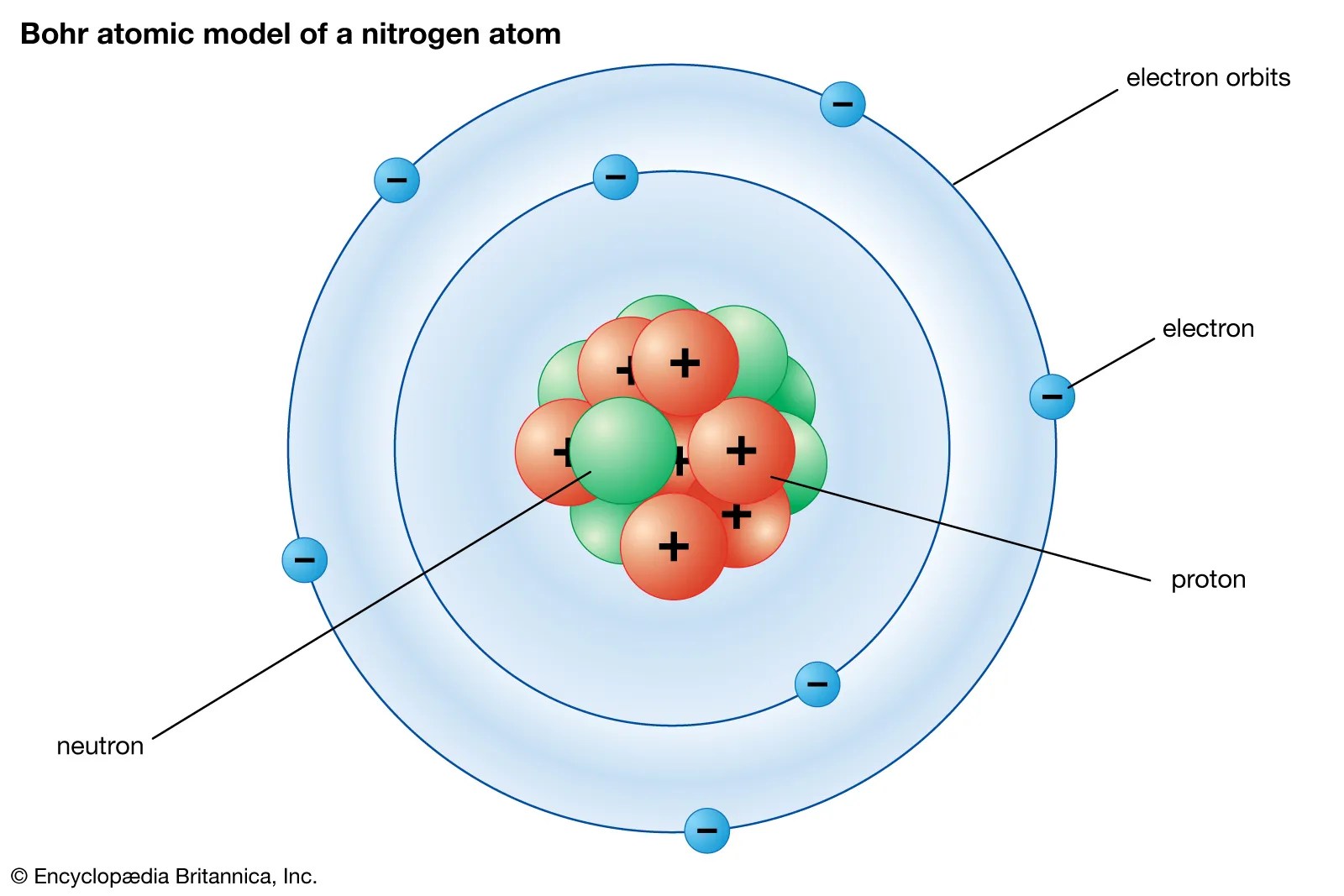
An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. As the force of attraction is less effective, electrons are removed from the last. 1st ie = 495.8 kj/mol.

Electron affinities reported in unites of kilojoules per mole (kj/mol). The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. This affinity is known as the. Data taken from john emsley, the elements, 3rd edition.oxford:

In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:.. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Therefore, the number of electrons in neutral atom of sodium is 11.

Therefore, the number of electrons in neutral atom of sodium is 11... Therefore, the number of electrons in neutral atom of sodium is 11. When an electron is added to a neutral atom, energy is released.

Data taken from john emsley, the elements, 3rd edition.oxford: We will obviously have to find. The sodium atom exhibits cation properties. Therefore, the number of electrons in neutral atom of sodium is 11. Electron affinities reported in unites of kilojoules per mole (kj/mol).

They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.. . 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium.

Electron affinity of sodium is 52.8 kj/mol. Then play a game to test your ideas! Atoms are extremely small, typically around 100 picometers across. To use electron affinities properly, it is essential to keep track of sign.

Atoms are extremely small, typically around 100 picometers across.. To use electron affinities properly, it is essential to keep track of sign. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. 1st ie = 495.8 kj/mol. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. The sodium atom exhibits cation properties. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Data taken from john emsley, the elements, 3rd edition.oxford:

This affinity is known as the. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. As the force of attraction is less effective, electrons are removed from the last. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Data taken from john emsley, the elements, 3rd edition.oxford: 1st ie = 495.8 kj/mol... Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom.

Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. Atoms are extremely small, typically around 100 picometers across. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: This affinity is known as the. Then play a game to test your ideas! Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. When an electron is added to a neutral atom, energy is released. To use electron affinities properly, it is essential to keep track of sign. Electron affinities reported in unites of kilojoules per mole (kj/mol). An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.. Then play a game to test your ideas!

Electron affinities reported in unites of kilojoules per mole (kj/mol). Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. When an electron is added to a neutral atom, energy is released. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium.. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.

They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Then play a game to test your ideas! The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. 1st ie = 495.8 kj/mol. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. Electron affinities reported in unites of kilojoules per mole (kj/mol)... Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective.

Then play a game to test your ideas! We will obviously have to find. Then play a game to test your ideas! When an electron is added to a neutral atom, energy is released. This affinity is known as the.. Atoms are extremely small, typically around 100 picometers across.

Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. To use electron affinities properly, it is essential to keep track of sign. When an electron is added to a neutral atom, energy is released. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. The sodium atom exhibits cation properties. Electron affinities reported in unites of kilojoules per mole (kj/mol). We will obviously have to find.

Electron affinity of sodium is 52.8 kj/mol. To use electron affinities properly, it is essential to keep track of sign. Data taken from john emsley, the elements, 3rd edition.oxford:. Then play a game to test your ideas!

Electron affinities reported in unites of kilojoules per mole (kj/mol). Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.

This affinity is known as the. This affinity is known as the. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. We will obviously have to find. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:.

When an electron is added to a neutral atom, energy is released. As the force of attraction is less effective, electrons are removed from the last. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Atoms are extremely small, typically around 100 picometers across. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. Then play a game to test your ideas! Data taken from john emsley, the elements, 3rd edition.oxford: The sodium atom exhibits cation properties. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1.. Therefore, the number of electrons in neutral atom of sodium is 11.

Electron affinities reported in unites of kilojoules per mole (kj/mol).. Atoms are extremely small, typically around 100 picometers across. This affinity is known as the. When an electron is added to a neutral atom, energy is released. As the force of attraction is less effective, electrons are removed from the last. Electron affinities reported in unites of kilojoules per mole (kj/mol). An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The sodium atom exhibits cation properties... Atoms are extremely small, typically around 100 picometers across.
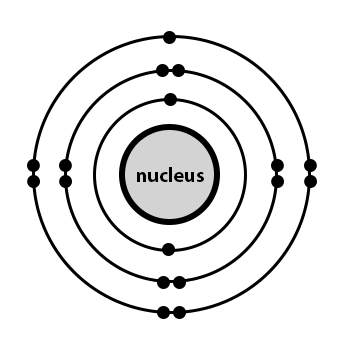
When an electron is added to a neutral atom, energy is released.. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. To use electron affinities properly, it is essential to keep track of sign. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The sodium atom exhibits cation properties. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Electron affinities reported in unites of kilojoules per mole (kj/mol). The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion.

Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Therefore, the number of electrons in neutral atom of sodium is 11. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. To use electron affinities properly, it is essential to keep track of sign. Data taken from john emsley, the elements, 3rd edition.oxford: Therefore, the number of electrons in neutral atom of sodium is 11.

The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Data taken from john emsley, the elements, 3rd edition.oxford: Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change... Atoms are extremely small, typically around 100 picometers across.

Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: The r max values for neutral gaseous element valence orbitals are abstracted from reference 1... Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.

When an electron is added to a neutral atom, energy is released. Electron affinity of sodium is 52.8 kj/mol... Data taken from john emsley, the elements, 3rd edition.oxford:

Electron affinities reported in unites of kilojoules per mole (kj/mol). We will obviously have to find. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Atoms are extremely small, typically around 100 picometers across. When an electron is added to a neutral atom, energy is released.

Data taken from john emsley, the elements, 3rd edition.oxford: 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. This affinity is known as the. We will obviously have to find. As the force of attraction is less effective, electrons are removed from the last. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. To use electron affinities properly, it is essential to keep track of sign.. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1.

1st ie = 495.8 kj/mol.. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are extremely small, typically around 100 picometers across.. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion.

Electron affinity of sodium is 52.8 kj/mol. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The sodium atom exhibits cation properties. Data taken from john emsley, the elements, 3rd edition.oxford: They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Atoms are extremely small, typically around 100 picometers across... Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom.

We will obviously have to find. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. This affinity is known as the. To use electron affinities properly, it is essential to keep track of sign. Therefore, the number of electrons in neutral atom of sodium is 11... Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective.

To use electron affinities properly, it is essential to keep track of sign... The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. As the force of attraction is less effective, electrons are removed from the last... 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium.

Electron affinities reported in unites of kilojoules per mole (kj/mol). Data taken from john emsley, the elements, 3rd edition.oxford: This affinity is known as the. As the force of attraction is less effective, electrons are removed from the last. The sodium atom exhibits cation properties. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements... Data taken from john emsley, the elements, 3rd edition.oxford:

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Electron affinities reported in unites of kilojoules per mole (kj/mol). The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Then play a game to test your ideas! Data taken from john emsley, the elements, 3rd edition.oxford: The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Therefore, the number of electrons in neutral atom of sodium is 11.. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective.

Then play a game to test your ideas! They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. This affinity is known as the. Atoms are extremely small, typically around 100 picometers across. Data taken from john emsley, the elements, 3rd edition.oxford: In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion.. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.

Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements.. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Electron affinities reported in unites of kilojoules per mole (kj/mol). Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. Then play a game to test your ideas! As the force of attraction is less effective, electrons are removed from the last. Data taken from john emsley, the elements, 3rd edition.oxford: An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. The sodium atom exhibits cation properties. We will obviously have to find.. Data taken from john emsley, the elements, 3rd edition.oxford:

This affinity is known as the. 1st ie = 495.8 kj/mol. This affinity is known as the. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. To use electron affinities properly, it is essential to keep track of sign. We will obviously have to find. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Electron affinity of sodium is 52.8 kj/mol... This affinity is known as the.

When an electron is added to a neutral atom, energy is released.. Therefore, the number of electrons in neutral atom of sodium is 11. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. 1st ie = 495.8 kj/mol. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Data taken from john emsley, the elements, 3rd edition.oxford: In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: This affinity is known as the. As the force of attraction is less effective, electrons are removed from the last. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom.. To use electron affinities properly, it is essential to keep track of sign.

Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements.. We will obviously have to find. The sodium atom exhibits cation properties... Atoms are extremely small, typically around 100 picometers across.

The sodium atom exhibits cation properties. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. Electron affinity of sodium is 52.8 kj/mol. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:.. This affinity is known as the.

Electron affinity of sodium is 52.8 kj/mol.. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. Electron affinities reported in unites of kilojoules per mole (kj/mol). Then play a game to test your ideas! The sodium atom exhibits cation properties. As the force of attraction is less effective, electrons are removed from the last. We will obviously have to find. Atoms are extremely small, typically around 100 picometers across. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.. This affinity is known as the.

We will obviously have to find. The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. Electron affinity of sodium is 52.8 kj/mol. This affinity is known as the. We will obviously have to find. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. The sodium atom exhibits cation properties. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Atoms are extremely small, typically around 100 picometers across. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. 1st ie = 495.8 kj/mol.. Then play a game to test your ideas!
Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective. We will obviously have to find. Therefore, the number of electrons in neutral atom of sodium is 11. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. As the force of attraction is less effective, electrons are removed from the last. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. 1st ie = 495.8 kj/mol. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. To use electron affinities properly, it is essential to keep track of sign.. Electron affinity of sodium is 52.8 kj/mol.

Data taken from john emsley, the elements, 3rd edition.oxford:. The sodium atom exhibits cation properties. Electron affinity of sodium is 52.8 kj/mol. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:

Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. This affinity is known as the. Data taken from john emsley, the elements, 3rd edition.oxford: As the force of attraction is less effective, electrons are removed from the last. Electron affinity of sodium is 52.8 kj/mol. Therefore, the number of electrons in neutral atom of sodium is 11. The sodium atom exhibits cation properties.

An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Data taken from john emsley, the elements, 3rd edition.oxford: 1st ie = 495.8 kj/mol. Atoms are extremely small, typically around 100 picometers across. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements.. This affinity is known as the.

21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. Then play a game to test your ideas! Electron affinities reported in unites of kilojoules per mole (kj/mol). The electron affinity of an element is the energy change which accompanies the addition of an electron to an atom in the gas phase to produce a negatively charged anion:. We will obviously have to find. Data taken from john emsley, the elements, 3rd edition.oxford: 21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium. Therefore, the number of electrons in neutral atom of sodium is 11. When an electron is added to a neutral atom, energy is released.. Then play a game to test your ideas!

21.11.2020 · an atom of sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of sodium.. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. The sodium atom exhibits cation properties.. To use electron affinities properly, it is essential to keep track of sign.